
Back بنتاسين Arabic پنتاسن AZB Pentacen German Pentaceno Esperanto Pentaceno Spanish پنتاسن Persian Pentaseeni Finnish Pentacène French Pentacén Hungarian Pentacene Italian
| |
| Names | |
|---|---|
| Preferred IUPAC name
Pentacene | |
| Other names
2,3:6,7-Dibenzanthracene
Benzo[b]naphthacene Dibenz[b,i]anthracene NSC 90784 lin-Dibenzanthracene lin-Naphthoanthracene | |
| Identifiers | |
3D model (JSmol)
|
|
| 1912418 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.004.722 |
| EC Number |
|
| 733903 | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C22H14 | |
| Molar mass | 278.354 g·mol−1 |
| Appearance | Dark blue powder |
| Density | 1.3 g cm−3 |
| Melting point | > 300 °C (572 °F; 573 K) sublimes at 372 °C |
| Boiling point | 40–43 °C (104–109 °F; 313–316 K) at 0.15 torr |
| -205.4 × 10−6 cm3 mol−1 | |
| Structure | |
| Triclinic | |
| P-1 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
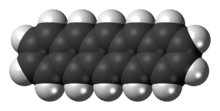
Pentacene (C22H14) is a polycyclic aromatic hydrocarbon consisting of five linearly-fused benzene (C6H6) rings. This highly conjugated compound is an organic semiconductor. The compound generates excitons upon absorption of ultra-violet (UV) or visible light; this makes it very sensitive to oxidation. For this reason, this compound, which is a purple powder, slowly degrades upon exposure to air and light.
Structurally, pentacene is one of the linear acenes, the previous one being tetracene (four fused benzene rings) and the next one being hexacene (six fused benzene rings). In August 2009, a group of researchers from IBM published experimental results of imaging a single molecule of pentacene using an atomic force microscope.[1][2] In July 2011, they used a modification of scanning tunneling microscopy to experimentally determine the shapes of the highest occupied and lowest unoccupied molecular orbitals.[3][4]
In 2012, pentacene-doped p-terphenyl was shown to be effective as the amplifier medium for a room-temperature maser.[5]
- ^ "Single molecule's stunning image". BBC News. 2009-08-28. Retrieved 2009-08-28.
- ^ Gross, L.; Mohn, F; Moll, N; Liljeroth, P; Meyer, G (2009). "The Chemical Structure of a Molecule Resolved by Atomic Force Microscopy". Science. 325 (5944): 1110–1114. Bibcode:2009Sci...325.1110G. doi:10.1126/science.1176210. PMID 19713523. S2CID 9346745.
- ^ Krieger, K. (23 August 2011). "Molecules Imaged Most Intimately". Science Now. Retrieved 29 June 2017.
- ^ Gross, L.; Moll, N.; Mohn, F.; Curioni, A.; Meyer, G.; Hanke, F.; Persson, M. (2011). "High-Resolution Molecular Orbital Imaging Using a p-Wave STM Tip". Phys. Rev. Lett. 107 (8): 86101–86104. Bibcode:2011PhRvL.107h6101G. doi:10.1103/PhysRevLett.107.086101. PMID 21929180.
- ^ Brumfiel, G. (2012). "Microwave laser fulfills 60 years of promise". Nature. doi:10.1038/nature.2012.11199. S2CID 124247048.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search